In our latest research project the Smithsonian Conservation Biology Institute and the Synthetic Biology Center Dept of Biological Engineering at MIT collaborated to test two technically challenging ideas using probiotic approaches to protect highly susceptible amphibians from the amphibian chytrid fungus (Bd). The paper was recently published in the Journal ISME Communications. We isolated a core frog skin bacteria that is found in high numbers on most golden frogs and genetically engineered it to produce an antifungal metabolite that kills the pathogen called violacein. By using a bacterium well-adapted to thrive on the frogs’ skin, that also produces antifungal metabolites we hoped to protect the frogs from disease. We were able to genetically modify one core skin microbe to produce violacein, but it did not persist well on the frog skin and was displaced after 4 weeks by the unmodified native bacteria strain. Treating the frogs with this genetically modified core skin microbe did not prevent the frogs from Bd or reduce infections.
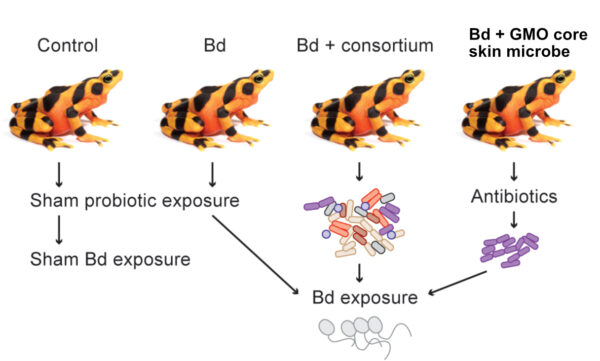
In this new experiment, we tested two new probiotic strategies to protect frogs from Bd. 1) Using a consortium of antifungal bacteria isolated from the frogs 2) Using a core skin microbe found on all golden frog skin that was genetically modified to produce antifungal metabolites.
In a second experimental group we mixed a consortium of seven antifungal bacteria that had been isolated from golden frog skin and supplemented the skin microbiome with these potentially beneficial microbes. Three of the seven bacteria persisted on the skin after 4 weeks, but this probiotic treatment also failed to protect the frogs from disease. While these results are disappointing, we were able to successfully test two technically-challenging ideas that have been discussed in the amphibian conservation community for many years. Furthermore, this research illustrates some of the challenges we still face in understanding and manipulating microbiomes and in using synthetic biology to solve real environmental problems.
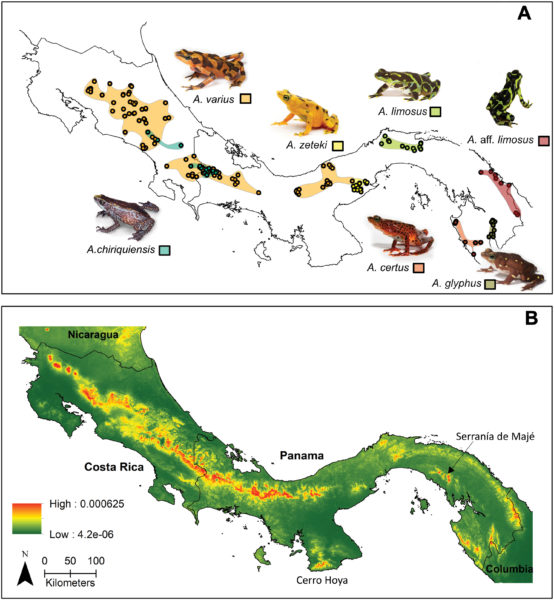
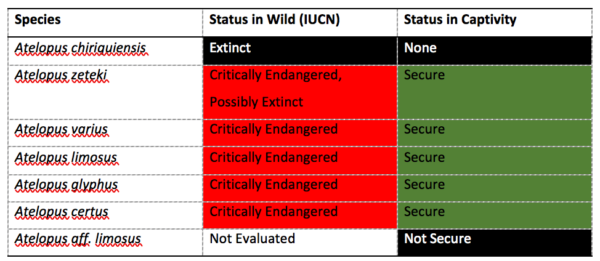
The research was led by Dr. Matthew Becker, Rob Fleischer and Brian Gratwicke (Smithsonian’s National Zoo and Conservation Biology Institute) and Dr. Jennifer Brophy and Christopher Voigt (Synthetic Biology Center Dept of Biological Engineering at MIT). Other collaborators include Ed Bronikowski, Matthew Evans, Blake Klocke, Elliot Lassiter, Alyssa W. Kaganer, Carly R. Muletz-Wolz (Smithsonian’s National Zoo and Conservation Biology Institute). Kevin Barrett (Maryland Zoo in Baltimore), Emerson Glassey & Adam J. Meyer (MIT). The work was funded by individual donors, the Smithsonian Institution Competitive Grants Program for Science, the Smithsonian Postdoctoral Fellowship, the U.S. Fish and Wildlife Service Division of International Conservation Amphibians in Decline Fund, the U.S. Defense Advanced Research Projects Agency’s Biological Robustness in Complex Settings program. The Maryland Zoo in Baltimore and the AZA Golden Frog Species Survival Program provided surplus-bred animals for research.
By Brian Gratwicke
Becker, M.H., Brophy, J.A.N., Barrett, K, Bronikowski, E., Evans, M., Glassey, E., Klocke, B. Lassiter, E., Meyer, A.J., Kaganer, A.W., Muletz-Wolz, C.R., Fleischer, R.C., Voigt, C.A., and Gratwicke, B. Genetically modifying skin microbe to produce violacein and augmenting microbiome did not defend Panamanian golden frogs from disease. ISME Communications